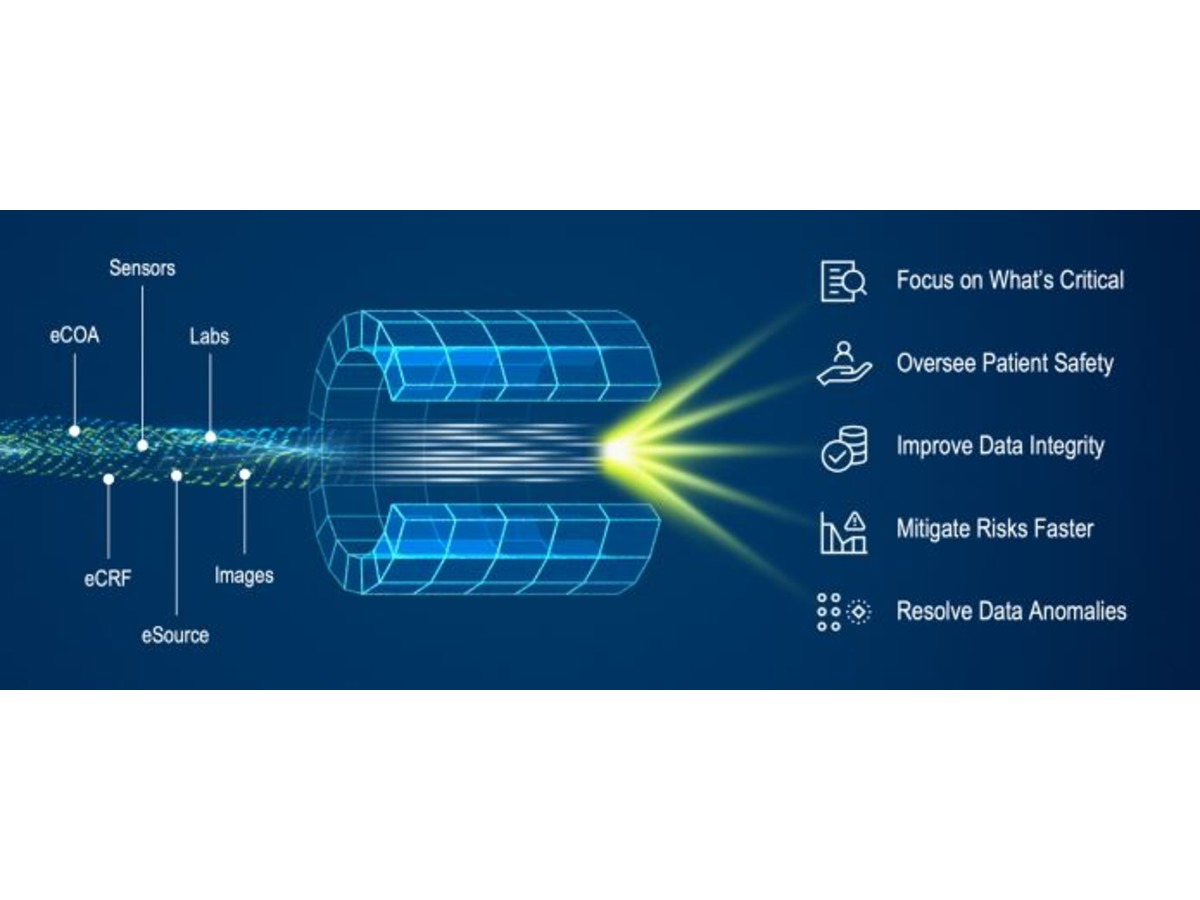
Medidata Detect: Ensure Quality With Comprehensive Data Surveillance and Risk Monitoring
By MedidataOversight of data quality and patient safety during a clinical trial is a team effort. The explosion of novel ways to collect and monitor on-site and remote data provides opportunities to uncover valuable insights during clinical research. However, it also increases the complexity and pressure on operational teams to support and scale these new approaches. Too often, siloed working groups and systems prevent timely identification and correction of quality issues, negatively impacting trial timelines and operational costs.
Download to find out more.

